How Roche cut SDTM creation time in half
Learn how Roche built governed R workflows that reduced SDTM dataset creation time by 50%, accelerating their clinical submission pipeline.

Proven results
Measurable outcomes in clinical analytics
Data science teams at firms like these ship faster, automate more, and give stakeholders direct access to the analysis behind their decisions.
50% faster SDTM creation
Roche's Insights Engineering team cut SDTM dataset creation time in half by moving clinical programmers from legacy tools to governed R environments on Posit Workbench.
R-based NDA accepted by the FDA
Novo Nordisk went from pilot R project to a complete R-based NDA submission accepted by the FDA in under two years, proving that open-source clinical programming meets regulatory expectations.
5x faster analysis
A Janssen scientist cut Alzheimer's disease risk marker analysis from 2.5 days to half a day after the team built RSVP, a scalable R infrastructure on AWS using Posit Connect and RStudio.
50% of clinical code migrated to R
GSK moved half of their clinical programming from SAS to R, reducing legacy licensing costs and giving their data scientists access to modern open-source tools on governed Posit environments.
Regulatory Compliance
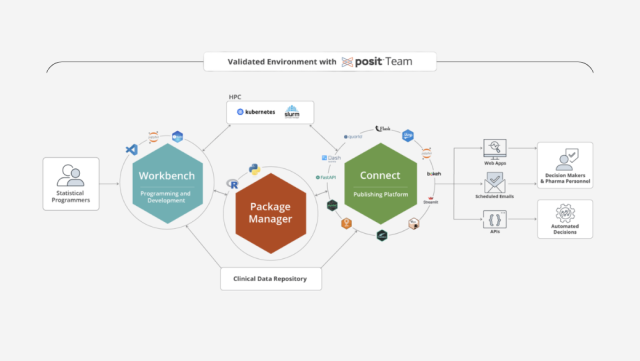
Run clinical analytics in governed environments
Develop, validate, and publish clinical analyses in R and Python with the traceability and reproducibility your regulatory and QA teams require.
How pharma teams build with Posit
Featured resources

Novo Nordisk: Journey to an R-based FDA submission

GSK: From Pilot Projects to Enterprise Adoption